[Uploaded by Carmel McNamara, IOS Press]
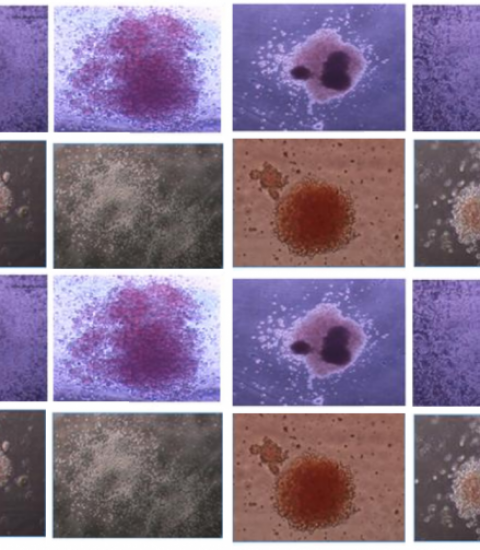
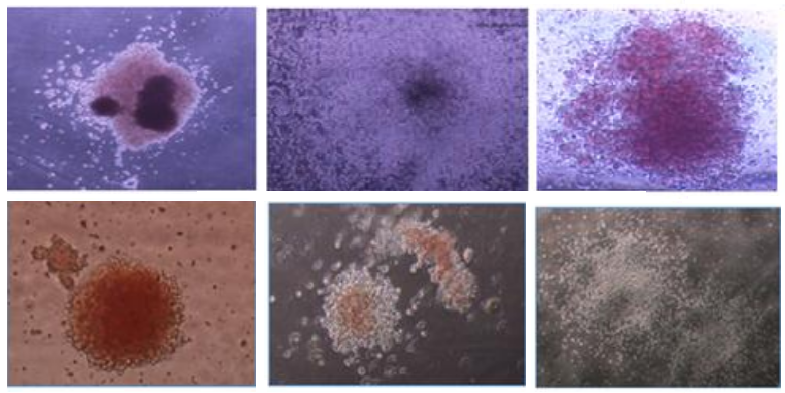
Amsterdam, NL – Just published in StemJournal is a study reporting that the earliest common hematopoietic and endothelial precursor designed as hemangioblast is highly expanded in stem cells assays using iPS cell lines derived from a patient with chronic myeloid leukemia. Patient-derived induced pluripotent stem cells (iPSC) represent a powerful tool to model several types of diseases and especially chronic myeloid leukema, and this article shows that chronic myeloid leukemia-derived iPSCs generate high numbers hemangioblasts detected by blast-cell colony forming cell (Bl-CFC) assays. Likewise, hematopoietic progenitors derived from either Bl-CFC or embryoid bodies are also highly increased in CML-derived iPSC.
This is an extract of the article published in the journal here
Chronic myeloid leukemia (CML) is the prototype of hematopoietic stem cell malignancy involving a highly primitive hematopoietic stem cell (HSC). The disease presents essentially by the expansion and proliferation of hematopoietic progenitors of myeloid lineage but lymphoid B cells lineage can also be involved [1]. There is a controversy with regard to the involvement of endothelial lineage in CML which would suggest that a stem cell with both hematopoietic and endothelial potential would be the target of transformation by BCR-ABL. Patient-derived induced pluripotent stem cells represent a powerful tool to model several types of diseases and especially chronic myeloid leukemia. We report here for the first time that the earliest common hematopoietic and endothelial precursor designed as hemangioblast is highly expanded in stem cells assays using iPS cell lines derived from a patient with CML. We show that CML-derived iPSCs generate high numbers hemangioblasts detected by blast-cell colony forming cell (Bl-CFC) assays. Likewise, hematopoietic progenitors derived from either Bl-CFC or embryoid bodies are also highly increased in CML-derived iPSC. Finally we show the potential of manipulating this expansion by using AHR pathway.
In this study, the control iPSC cell line (PB33) was obtained using hematopoietic cells of a normal donor using the same protocol. All analyses were performed using a polyclonal stock of iPSC. Both cell lines were characterized using in vitro and in vivo pluripotency tests.
Discussion
The elusive nature of the most primitive CML stem cells which are very difficult to study in human samples stimulated a major research during the last 10 years to generate models of CML stem cells. As matter of fact, primary CML stem cells which are very difficult to isolate and expand, are at the origin of relapses upon discontinuation of tyrosine kinase inhibitors (TKI) therapies. Several murine CML models have been developed to this purpose but they do not recapitulate faithfully the most primitive human CML cells. The use of embryonic stem cell models is of interest as the overexpression of BCR-ABL has been shown to be a major stimulus for generation of long-term leukemic hematopoiesis from murine embryonic stem cells (ESC). However, one of the limitations of the ESC models is the need to overexpress BCR-ABL under the control of different promoters potentially leading to results not relevant to the pathophysiology of human CML. With the advent of iPSC technology, it became of major interest to use human CML cells for reprogramming with the goal of reproducing the earliest stages of human CML and potentially to use them for drug screening purposes.
The iPS cell line used in the experiments reported here was generated from the primary leukemic cells of a 14-year old patient with TKI-resistant leukemia. The first goal of the reprogramming of the leukemic cells of the patient was to determine if BCR-ABL expression and induced signaling could be intact at different stages. We show in this study that BCR-ABL expression is present at the most primitive iPSC stage and BCR-ABL autophosphorylation is present in this context as it would have been expected in an adult leukemic cell context.
View methods and discussion in the published article!




