Guidelines for Submitting Protocols
![]() Submissions are welcomed via Editorial Manager. Click on the button to send us your manuscript. First, please read the guidelines carefully.
Submissions are welcomed via Editorial Manager. Click on the button to send us your manuscript. First, please read the guidelines carefully.
New protocols or previously published protocols having significant changes to them will be published in StemJournal following peer review and acceptance. Furthermore, these protocols will be archived in StemBook (see here), while retaining the original StemJournal citation. Minor updating, of protocols which are already published in StemBook, will be published following peer review and acceptance as protocol updates directly in StemBook.
Open Access Journal
All content will be published Open Access (OA). Each protocol will be freely available from the moment it is published, also in the pre-press module. For the first volumes, the article processing charges (APCs) are significantly reduced (by 80%) to an affordable US$ 300 / € 260 per published paper. Invited articles are exempt from the APC.
Click here for further details about APCs.
Click here for more information about the IOS Press Open Library® policy, and also to check if there are any institutional agreements that you might be eligible for.
Preparation of Manuscripts
Protocols can be submitted for review or commissioned by any one of the Editors or Associate Editors. Submitted protocols will undergo peer review. Protocols, which are commissioned, will also undergo peer review and will only be published on acceptance.
Organization and style of presentation
- Manuscripts must be written in US English. Authors whose native language is not English are recommended to seek the advice of a native English speaker or English language service before submitting their manuscripts.
- Manuscripts should be double spaced throughout with wide margins (2.5 cm or 1 in), including the “Abstract” and “References.” Every page of the manuscript, including the title page, references, tables, etc., should include a page number centered at the bottom. Do not number headings or subheadings.
- There are no page or word limits for protocol submissions but manuscripts over 10,000 words (“Introduction” through “Discussion”) should be approved by the Editors-in-Chief before submission
Title page
- Title (should be clear, descriptive, concise, and avoid the use of abbreviations)
- Full name(s) of author(s)
- Full affiliation(s). Delineate affiliations with lowercase letters.
- Present address of author(s), if different from affiliation
- Running title (45 characters or less, including spaces)
- Complete correspondence address, including telephone number, fax number and email address
- Leave the author information blank if double-blind peer review is wished for, but do include the information in the cover letter
Authorship
To be considered as an author of an article, the following criteria must be met:
- Substantial contributions to the conception or design of the work; or the acquisition, analysis, or interpretation of data for the work; and
- Drafting the work or revising it critically for important intellectual content; and
- Final approval of the version to be published; and
- Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
When submitting the manuscript, the author listing and order should be final. If any addition, deletion, or rearrangement of author names in the authorship list does need to be made after submission, this can be done only before acceptance and with the Editor’s approval. To request such a change, the Editor must receive the following from the corresponding author: (1) the reason for the change in author list and (2) written confirmation from all authors, including the affected author, that they agree with the addition, removal, or rearrangement. Only in exceptional circumstances will the Editor consider the addition, deletion, or rearrangement of authors after the manuscript has been accepted. While the Editor considers the request, publication of the manuscript will be suspended. If the manuscript has already been published in an issue, any requests approved by the Editor will result in an Erratum. Please contact the Editorial Office (stemjournal@iospress.com) for more information.
If any author is also a member of the StemJournal Editorial Board at the time of article submission, this should be declared in the Conflict of Interest Section (see instructions in the section for Conflict of Interest, below).
Abstract and Keywords
- The abstract should be concise, clear, descriptive, self-explanatory, and no longer than 250 words (do not include headings)
- Please follow the suggested structured format: Introduction (briefly summarizing the protocol), General Procedure, Expected Results (a maximum of two sentences), Timing (one or two sentences describing the timing)
- Include a list of 4–10 keywords. These keywords should be terms from the MeSH database.
Introduction
Provide enough information to put your work into context. Be concise. Clearly address the following points:
- What information is already available?
- What is the rationale or reason for your research?
- What problem(s) does it address?
Do not include a comprehensive literature review of research related to the protocol but do cite relevant papers where (a version of) the protocol had been described previously. End the Introduction by clearly describing the suitability of the protocol for specific research.
Workflow
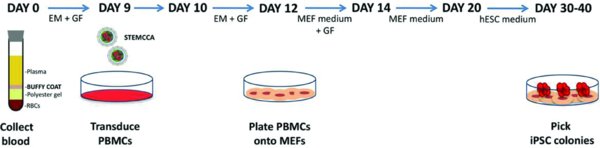
Submit a clear, visual workflow of the protocol. Include major steps for the protocol, for example:
From: Gianotti-Sommer, A., Rozelle, S.S., Sullivan, S., Mills, J.A., Park, S.-M., Smith, B.W., Iyer, A.M., French, D.L., Kotton, D.N., Gadue, P., Murphy, G.J. and Mostoslavsky, G., Generation of human induced pluripotent stem cells from peripheral blood using the STEMCCA lentiviral vector (April 29, 2013), StemBook, doi/10.3824/stembook.1.93.1 (link).
Materials and Preparation
This section should be tabulated, well-structured and detailed enough for others to be able to know what materials are required. Use clear subheadings throughout, such as: "Isolation of Brain Cells," "Culturing Neurons," "Immunocytochemistry," "Antibodies" etc. For example, see the table listed under section 4 (Materials) here.
For antibodies, make sure to include the sourcing of antibodies used accurately. You can use subheadings, such as “Equipment,” “Reagents,” “Andibodies,” etc. List any essential information underneath the subheading. Hazardous chemicals and reagents should be labeled as such, with an additional bracketed comment, such as: “(USE CAUTION)”. Make sure to include company or supplier name and location (city, state, country), web address and catalog number.
Preparation
This section should be a bulleted list and start with describing any preparations necessary, such as preparation of any medium used or reagent, and should have clear subheadings, for example: "Neural Differentiation Medium," "Astrocyte Differentiation Medium," "Oligodendrocyte Differentiation Medium," etc.
Refer to the following protocol format and structure as a guide:
Yang, W., Mills, J.A., Sullivan, S., Liu, Y., French, D.L., and Gadue, P., iPSC Reprogramming from human peripheral blood using sendai virus mediated gene transfer (December 11, 2012), StemBook, ed. The Stem Cell Research Community, doi/10.3824/stembook.1.73.1 (link).
[Note to authors: The published protocols in StemBook referenced here should be used as ‘guides’ only. Authors are requested to follow these “Guidelines for Submitting Protocols” as closely as possible.]
Use the “active voice” and present tense when describing the preparation. For example: “To prepare 100 ml of oligodendrocyte differentiation medium, aseptically mix the following components. For larger volumes, increase the component amounts proportionally. If desired, add 1 ml of antibiotic-antimycotic solution per 100 ml of medium.” (example text reproduced with permission from Life Technologies™)
Present all components used in the preparation of the medium in tabular format, including the “Component,” “Final Concentration,” and “Amount,”
Preparation of Media
This is the major part of the protocol and must be a bulleted list, ideally with numbers in bold; do not follow the numbers with a period (full stop). Use the active tense rather than the passive tense, for example: "Pipette 20 ml of buffer A into the flask" (and not: "20 ml of buffer A are/were pipetted into the flask"). If the protocol naturally breaks into separate stages, then include subheadings and resume the numbered list. Include a “Timing” callout with each subheading and state how long the section will take to complete. Subheadings are particularly appropriate after steps in the protocol where the procedure can be stopped (pause point), i.e. when the experiment can be stopped and resumed at a later date. Any pause points should be indicated with the heading “Pause Point,” followed by a brief description of the options available, for example: "Can be left overnight at 4 °C or frozen for up to a month at –20 °C."
Highlight critical steps in the protocol that must be performed in a very precise manner, e.g., where the time and temperature of a step is crucial or the use of RNase free solutions is required, thus providing the user with hints to maximize the likelihood of success. Make these clear with the heading “Critical Step,” followed by a brief explanation.
Highlight any toxic or harmful chemicals that are used. Make these clear by preceding them with the wording “CAUTION” prior to their first mention and include brief details of the hazard and the appropriate handling information. Include diagrams and/or photographs of equipment set-up, where appropriate. If the protocol is complicated, you should consider including a flow diagram to demonstrate how the stages fit together. Movies of particularly complicated procedures can be submitted.
Where there are alternative routes to reach the next stage of the protocol, please give enough background so that the reader will be able to make an informed decision on the route to choose. Letters of the Latin alphabet [(A), (B), (C)] should be used to identify the different options, and Roman numerals [(i), (ii), etc.] should be used to break down the appropriate steps. These options cannot have subheadings.
Please state all centrifugation speeds in g and include the length of time and temperature of the centrifugation, for example: "Centrifuge at 14,000 g for 5 min, 4 °C."
Please include “Troubleshooting” callouts after steps where problems are encountered, that are subsequently mentioned in the Troubleshooting section.
Timing
Please include a timeline showing the time each step takes. You can also group a series of steps together, for example: “Steps 1–3, 20 min; Steps 4+5, 1 h.” The timeline should be in the form of a bulleted list and follow each procedure. Always keep in mind the reproducibility of the steps.
Expected Results
Comment on the expected results which can be regularly achieved with this protocol, and/or results that may be produced from other applications or experimental conditions than those used from the protocol.
Troubleshooting
Include information on how to troubleshoot the most likely problems users will encounter with the protocol. Please provide this information in the form of a table with the columns: “Step”, “Problem”, “Possible Reason,” and “Solution”. The step number should be given where the problem is first observed (not where it occurred). The appropriate steps should also be flagged in the main text by adding “Troubleshooting” callouts. If troubleshooting text refers to only one or two steps, it can also be formatted as normal text with subheadings referring to the steps or sections that the information pertains to.
Acknowledgements
Include individuals or companies which have assisted with your study, including advisors, administrative support and suppliers who may have donated or given materials used in the study. If there are none, include the section and insert: "The authors report no acknowledgments."
Funding
Include all funding sources. If there are none, include the section and insert: "The authors report no funding."
Conflict of Interest
If there is no conflict of interest to declare, include this section and insert: "The authors report no conflict of interest." If an author is on the Editorial Board of StemJournal at the time of submission, the following statement should be included in this section: "<AUTHOR> is an Editorial Board Member of StemJournal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.’"
References
Authors are requested to use the Vancouver citation style. Place citations as numbers in square brackets in the text. All publications cited in the text should be presented in a list of references following the text of the manuscript. Only articles published or accepted for publication should be listed in the reference list. Submitted articles can be listed in the text as [author(s), unpublished data]. All authors should be listed in the reference list. If an article has a DOI, this should be provided after the page number details. The number is added after the letters “doi.” Manuscripts will not be considered if they do not conform to the Vancouver citation guidelines.
References must be listed in Vancouver style:
[1] Alzheimer Research Forum, Drugs in Clinical Trials: AAB-001, http://www.alzforum.org/drg/drc/detail.asp?id=101, Last updated May 29, 2007, Accessed on January 29, 2008.
[2] Smith M.A. (2006) Oxidative stress and iron imbalance in Alzheimer disease: how rust became the fuss! In Alzheimer's Disease: A Century of Scientific and Clinical Research, Perry G, Avila J, Kinoshita J, Smith MA, eds. IOS Press, Amsterdam, pp. 305–308.
[3] Hendriks, W.T., Warren, C.R., Cowan, C.A. (2016) Genome Editing in Human Pluripotent Stem Cells: Approaches, Pitfalls, and Solutions. Cell Stem Cell 18, 53–65.
[4] Li, L. Xie, T. (2005). Stem cell niche: structure and function. Annu Rev Cell Dev Biol 21, 605–631.
[5] Swanson, L.W. (1992) Brain maps: structure of the rat brain, 240 pp. ISBN: 0444814140, Elsevier, Amsterdam.
[6] Swanson, L.W. (2004) Brain maps: structure of the rat brain. A laboratory guide with printed and electronic templates for data, models and schematics, 3rd revised edition, 215 pp. with CD-ROM, Brain maps: computer graphics files 3.0. ISBN: 0126105820, Elsevier, Amsterdam.
[7] Babiarz, J.E., and Blelloch R., Small RNAs – their biogenesis, regulation and function in embryonic stem cells, StemBook, ed. The Stem Cell Research Community, StemBook (http://www.stembook.org/), doi/10.3824/stembook.1.47.1, Posted 1 May 2009, Accessed 29 May 2009
Datasets and Data Articles
- All datasets and data articles referenced in your manuscript should be cited in the main reference list of your article (not in a separate box or in the article text)
Tables
- Number according to their sequence in the text. The text should include references to all tables.
- Provide each table on a separate page of the manuscript after the references
- Include a brief and self-explanatory title with any explanations essential to the understanding of the table given in footnotes at the bottom of the table
- Vertical lines should not be used to separate columns. Leave some extra space between the columns instead.
- Citations in the tables should be numbered and included in the “References”
Figures Legends/Figures
- Number the figures according to their sequence in the text. The text should include references to all figures.
- Each figure should be provided on a separate page, not included in the text
- Figures should preferably be formatted in TIF or EPS format. JPG is also acceptable.
- Composite figures must be preassembled
- Figures should be designed with the format of StemJournal in mind. They should be of such a size as to allow a reduction of 50%.
- Line art should have a minimum resolution of 1200 dpi and be saved as an EPS or TIF:
- Do not use faint lines and/or lettering and check that all lines and lettering within the figures are legible at the final size
- All lines should be at least 0.1 mm (0.3 pt) wide
- Vector graphics containing fonts must have the fonts embedded in the files
- Grayscale figures (including photos) should have a minimum resolution of 300 dpi, or 600 dpi for combination art (lettering and images) and be saved as a TIF
- Figures should be cropped to include the figure only (no blank space)
- Do not save line art as JPG; this format may lose information in the publishing process
- Do not use figures taken from the Internet; the resolution will be too low for printing
- For figures to be printed in color, please send a CMYK encoded EPS or TIF
- On figures where a scale is needed, use bar scales rather than numerical ones, i.e., do not use scales of the type 1:10,000. This avoids problems if the figure needs to be reduced.
- Each figure should have a self-explanatory caption, which should be typed separately from the figure in the manuscript
- Photographs are only acceptable if they have good contrast and intensity.
- Color figures will be published online at no charge
- Costs for color figures in the print version of the journal are as follows: 1 figure – 650 euro; 2 figures – 900 euro; 3 figures – 1050 euro; 4 figures – 1200 euro; 5 figures – 1350 euro. Cost for each additional color figure will be 150 euro. Unless the color printing charge is paid, color figures will be automatically adjusted to grayscale in print. You may opt to send in both black/white figures for print, and color figures for the online PDF (please adjust the figure legend appropriately)